AstraZeneca (AZN) said its Farxiga diabetes drug is the first inhibitor to receive approval by the U.S. Food and Drug Administration (FDA) to be used for the treatment of heart failure.
“With the approval ofFarxiga, we have reached a critical milestone to potentially transform heart failure treatment for the millions of people living with the condition in the US,” said Mene Pangalos, AstraZeneca Executive Vice President of BioPharmaceuticals R&D.“We are now one step closer to making a significant impact on their lives by providing a much-needed treatment to help reduce their disease burden and live longer.”
Heart failure is a life-threatening disease in which the heart cannot pump enough blood, affecting about 64 million people worldwide – at least 50% have a reduced ejection fraction – and six million in the US.
The FDA approval followed “positive results” from the landmark Phase III trials, which showed that Farxiga led to a statistically significant and clinically meaningful reduction of cardiovascular death or hospitalisation for heart failure, compared to placebo, AstraZeneca said. During the trial duration, one cardiovascular death or hospitalisation for heart failure could be avoided for every 21 patients treated with Farxiga.
“Today’s approval provides physicians with a completely novel pharmacological approach that greatly improves outcomes for patients with heart failure with reduced ejection fraction,” said John McMurray from the Institute of Cardiovascular and Medical Sciences at the University of Glasgow.
The drugmaker’s shares rose 2% to $54.55 in midday U.S. trading.
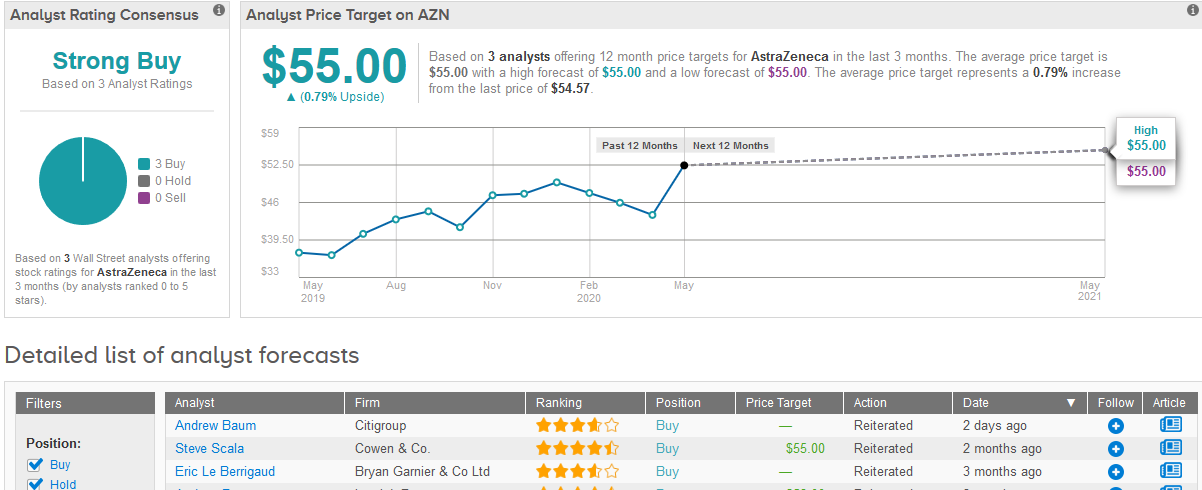
TipRanks data shows that the Wall Street analysts covering the stock in the past three months have a bullish outlook awarding a Strong Buy consensus rating. The $55 average price target suggests very limited upside potential of less than 1% in the coming 12 months. (See AstraZeneca stock analysis on TipRanks).
Related News:
Gilead in Talks to Ramp Up Global Supply of Remdesivir Production
Incyte Reports Strong Jakafi Sales, But Recent Rally Sparks Downgrades
SEC Puts the Kibosh on 3 Biotech Stocks Hawking a Miracle Anti-Coronavirus Drug